Reviewed by the clinical team at Renew Bariatrics, a medical tourism practice in Mexico specializing in bariatric surgery for patients from the U.S. and Canada. Our surgeons have performed thousands of weight-loss procedures and work with patients whose genetic and metabolic profiles make sustained weight loss especially difficult.
Over decades, weight management has been characterized by a “one-size-fits-all” philosophy. Cliched mantras such as “calories in/calories out” and “eat less, move more” have formed the foundations of innumerable programs, often accompanied by a revolving carousel of fad diets, such as low-fat, keto and paleo. But in spite of this plethora of advice, a nagging, deeply personal question lingers: How can an eating plan that miraculously works for one individual have very little, or no, effect on another?
The answer to this maddening problem is becoming all too plain. We’ve simply been disregarding the most simple instruction manual we have: Our unique genetic codes. Top health experts across the world, including the World Health Organization (WHO), acknowledge that a complex association between genetic, behavioral and environmental factors contributes to obesity and metabolic health, instead of individual discipline by itself.
We are currently moving into a game-changing phase of personal health, one that replaces strict, generalized prescriptions with a plan that suits personal biology. This change does not dismiss the accepted premises of good health, but refines them. Contemporary DNA testing and the study of family health history are two powerful tools that can be used to enhance individual health. When combined, they can help to form a more specific, human and sustainable weight management framework, one that does not oppose biological diversity, but lives within it. In our own clinical experience at Renew Bariatrics, patients often arrive after years of frustration with diets that worked for everyone but them — a pattern that frequently traces back to the genetic and hormonal factors described below.
The Grand Canyon Between Theory and Practice: Why Generic Plans Fail
The underlying principle of weight management is the indisputable scientific principle of energy balance: If you consume fewer calories than you use, you’ll lose weight. But according to the National Institutes of Health (NIH), there are vast differences in the way individuals consume and expend energy. One of the main culprits? Genetics.
Genes play a role in many factors surrounding weight loss, including metabolism rate, insulin sensitivity, hunger hormones, fat oxidation, muscle growth and even automatic preferences toward food. Two people could have the same diet and exercise regimen, be very careful about counting calories, and have very different results.
This has been confirmed by recent large-scale, genome-wide association studies, which have found that specific genetic variants may be responsible for differences in how macronutrients are processed by the human body. For example, a meta-analysis published in the American Journal of Clinical Nutrition found that variants in the FTO gene on chromosome 16 were associated with higher protein intake, independent of body mass index.
Consider dietary response. An individual can thrive with a high-carbohydrate, plant-based diet, enjoying stable blood sugar, little hunger and regular fat loss. Another, consuming the same food, can have cravings, be tired, and lose no weight at all. One factor that may play a role is a common variant near the FGF21 gene on chromosome 19 called rs838133.
Analyses of more than 450,000 people have shown that carriers of the minor A allele tend to consume a higher percentage of carbohydrates and less protein and fat, likely because the variant alters signaling by fibroblast growth factor 21 (FGF21) — a hormone that helps regulate sugar preference and the metabolism of glucose and fats.
The same happens with exercise response. Some people get strong and build muscle fast through resistance training, while others realize more fat loss and cardio gains through endurance-based programs and exercises.
These disparities, then, don’t point to a lack of motivation or discipline. They are manifestations of genetic predisposition. Findings like these have given rise to the explosive growth of nutrigenomics and exercise genomics — disciplines that focus on gene-nutrition and gene-physical activity interactions. Many large-scale studies have noted how genetic variations can alter individual reactions to food, specific exercises and length of exercise sessions.
For example, a study published in AIMS Molecular Science in 2024 found that obese individuals who followed a nutrigenetic diet customized for their genetic makeup lost significantly more weight than those who followed a generic diet. The nutrigenetic group also lost more body fat and experienced improved triglyceride levels.
Another study published in BMC Nutrition in 2020 found that while a ketogenic diet produced more weight loss in the first six months, overweight and obese individuals following a personalized nutrigenetic diet lost significantly more weight over an 18-month follow-up — and saw greater improvements in cholesterol and fasting glucose.
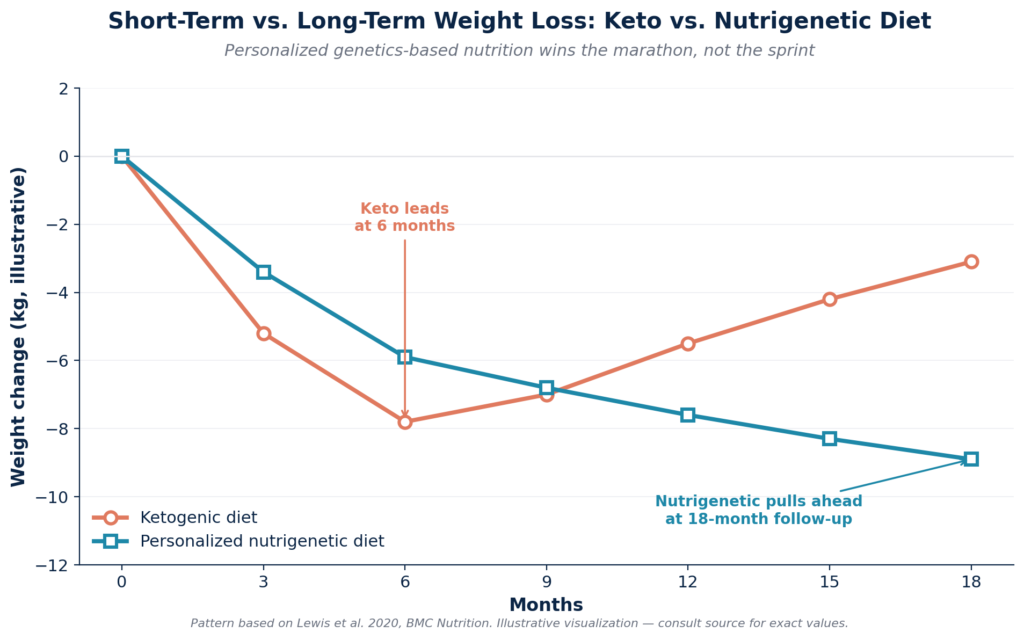
The new school of thought is evident: genetics do not determine fate, but they do have an impact on efficiency. When genetic factors are not considered in weight loss, frustration and burnout can result. But when they’re honored and taken into account, weight loss becomes more aligned with the body and easier to sustain.
Cracking the Playbook: What the Contemporary DNA Tests Can and Cannot Reveal
Many contemporary consumer tests analyze single nucleotide polymorphisms, or SNPs, which are small genetic variations that may be associated with differences in appetite, metabolism, exercise response, and recovery. Used carefully, these tests can offer insight into biological factors rather than fixed outcomes. A person may carry variants associated with traits such as hunger regulation, insulin sensitivity, caffeine metabolism, aerobic capacity, or recovery patterns. In that sense, genetic testing can sometimes help explain why individuals do not always respond the same way to identical diet or training strategies.
Still, the limits are important. These tests cannot determine with certainty why a person has gained weight, predict exactly which nutrition plan will work best, or fully capture how someone will perform in everyday life. Traits related to body weight and fitness are shaped by a wide mix of influences, including many genes, along with sleep, stress, environment, medications, and daily habits.
For that reason, contemporary DNA tests are best understood as one source of information rather than a definitive roadmap. They may provide useful context, and in some cases meaningful direction, but they should be interpreted with caution and alongside the broader behavioral and physiological picture.
Hormonal Signaling and Appetite Regulation
Here’s the thing. Hunger is not always just about self control, and honestly, that matters more than people like to admit.
Some genes, including FTO, seem to shape how hungry a person feels, how full they feel after eating, and even how rewarding food feels in the first place. One of the better known variants, rs9939609, has been linked to a higher risk of obesity and appears to affect appetite regulation through changes in fullness signaling after meals and ghrelin-related responses. So, in plain English, some people may get hungry faster, feel less satisfied after eating, or just have a tougher time pumping the brakes around food. Not because they’re weak. Just because biology does not make it easy.
And then, of course, you drop that biology into modern life, where ultra-processed food is everywhere, portions are huge, and snacks are basically engineered to keep calling your name, and it can become a real uphill battle.
That doesn’t mean anyone is doomed. It also doesn’t mean the issue is character. What it means, more likely, is that some people need tighter strategies because their internal hunger cues are a little less cooperative. More protein can help. Fiber too, when used in a way that actually works in real life. Structured meals, fewer food triggers, less random grazing, that sort of thing. Not glamorous, maybe, but effective.
Adaptation and Capacity to Exercise
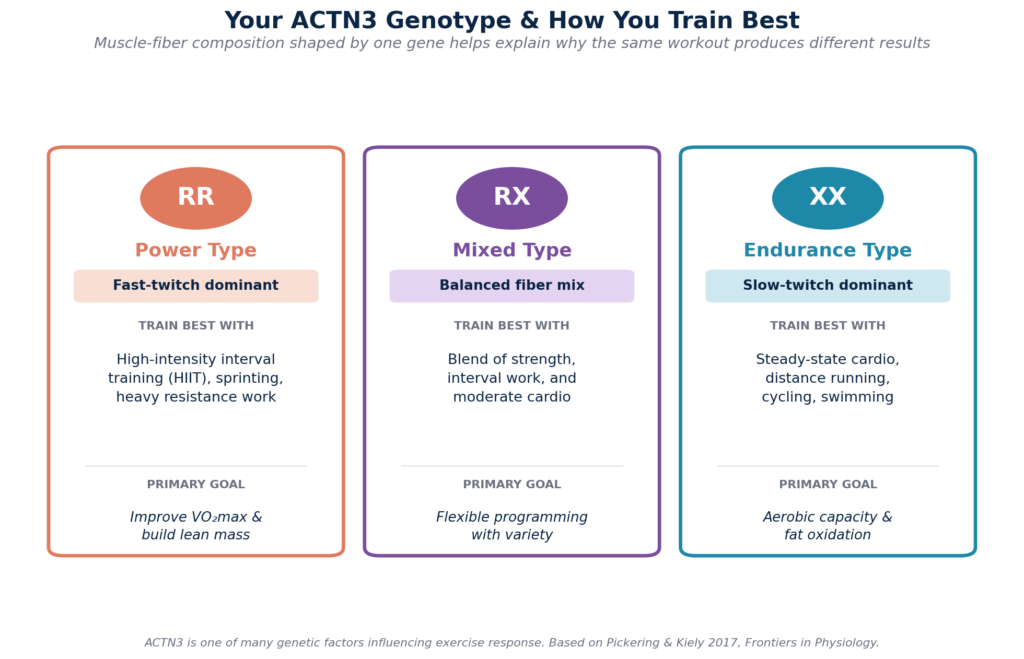
Genes such as ACTN3 also affect the composition of the muscles, which means that power-based exercises may come more naturally to some people, while others adapt more easily to endurance-based activities. The RR genotype enhances fast-twitch muscle fibers within the body, which are better for sprinting and other types of high-intensity training, whereas the XX genotype tends to have more slow-twitch endurance fibers that are better for gaining aerobic benefits through steady cardio exercises.
The International Sports Sciences Association (ISSA) provides educational material that outlines the impact of genetic variation on an individual’s potential strength and aerobic capacity, as well as their recovery requirements. Aligning training with these tendencies tends to enhance fitness outcomes and decrease the risk of injuries and mental exhaustion.
Absorption and Utilization of Micronutrients
Differences in genes may disrupt the absorption or release of such nutrients as Vitamin D, B12, folate, magnesium and iron. The NIH Office of Dietary Supplements maintains fact sheets on each of these nutrients and the impact of deficiencies on metabolism, hormonal equilibrium and energy generation. Correcting these deficiencies can meaningfully enhance weight-management results.
Notably, DNA tests do not make predictions, but provide only estimates. The information they provide is about inclinations, not guarantees. According to the National Library of Medicine, genetic risks are affected by lifestyle decisions and can be modulated through epigenetic mechanisms — the ways cells affect gene activity without altering the underlying DNA. In other words, you can have a specific genetic tendency, but it may not become apparent until certain lifestyle choices bring it to the surface.
The Living Legacy: Family History as a Critical Health Lens
Family history has been the main predictor of disease risk for far longer than genetic testing has existed. Family patterns show the behavior of shared genetics over time in real-life situations. According to the Centers for Disease Control and Prevention, multiple genes contribute to a person’s feelings of hunger, sense of fullness, and metabolism — which is why obesity, diabetes, thyroid disorders, PCOS, cardiovascular disease, and weight gain around menopause all tend to run in families.
For example, if several family members are shown to have insulin resistance despite low-carb diets, it could mean that they all possess a similar variant in insulin pathway genes. This might mean that fiber-enriched carbs could be a better choice for weight loss in these people.
In addition, cultural and environmental factors such as traditional diets, activity levels, stress exposure, and socioeconomic conditions can affect gene expression through many generations.
The Genetics of Severe Obesity: When Biology Outpaces Lifestyle
For most people, nutrigenetic and lifestyle strategies are enough. But when variants in FTO, MC4R, and leptin-pathway genes stack with environmental pressures, the result can be severe, treatment-resistant obesity — the kind where hunger signaling, energy expenditure, and fat storage are all working against the patient simultaneously.
This is the population for whom clinical guidelines recommend considering bariatric surgery: adults with a BMI of 40 or higher, or 35+ with obesity-related conditions like type 2 diabetes, sleep apnea, or hypertension. Surgery is not a shortcut around genetics; it’s a way of changing the hormonal conversation so that lifestyle changes can finally take hold.
From Insight to Action: Developing a Dynamic Personalized Plan
Genetic insight is only really powerful when put into practice. According to a study published in Nature Medicine, precision nutrition and fitness convert abstract data into actionable decisions. This reflects the rising scientific trends of precision medicine and precision nutrition.
Nutrition Strategy
Rather than strict diets, people should develop a structure of diet that promotes a healthy metabolism. The macronutrient ratios, meal schedule, fiber intake, and protein requirements are modified according to genetic response and lifestyle needs.
For example, carriers of FTO risk alleles tend to show higher baseline levels of the hunger hormone ghrelin and lower satiety-hormone leptin, which may help explain why they feel hungrier more often and often benefit from meal structures that prioritize protein and fiber. Carriers of common FGF21 variants may respond differently to varying carbohydrate loads because the same variants shape sugar preference and macronutrient balance. Supplementation can also be focused rather than speculative — for example, magnesium may be prioritized for those with genetic variants that affect its absorption.
Fitness Design
Training should be consistent with one’s recovery capacity, risk of injury, and tendencies toward specific types of exercise. For example, ACTN3 R allele carriers may benefit from high-intensity interval training (HIIT) to improve VO₂max, while XX genotype individuals may respond better to moderate, steady-state cardio. Genetics can affect exercise quantity and enjoyment, as well as capacity for recovery from activity.
Environmental Design and Behavior
The psychological shift is, perhaps, the most radical when it comes to the role of genetics in health. Knowledge about biological predisposition decreases shame and redefines problems as variables that can be solved. Adjusting environments to achieve weight loss success — such as practicing good sleep hygiene, having nutritious food available, coping with stress in a healthy way, and establishing good health habits — can also make people less solely reliant on willpower-based strategies. Apps that combine DNA results with habit-tracker mechanisms could make it easier to approach weight loss from both behavioral and genetic standpoints.
Surgical Options When Genetics Stack the Deck
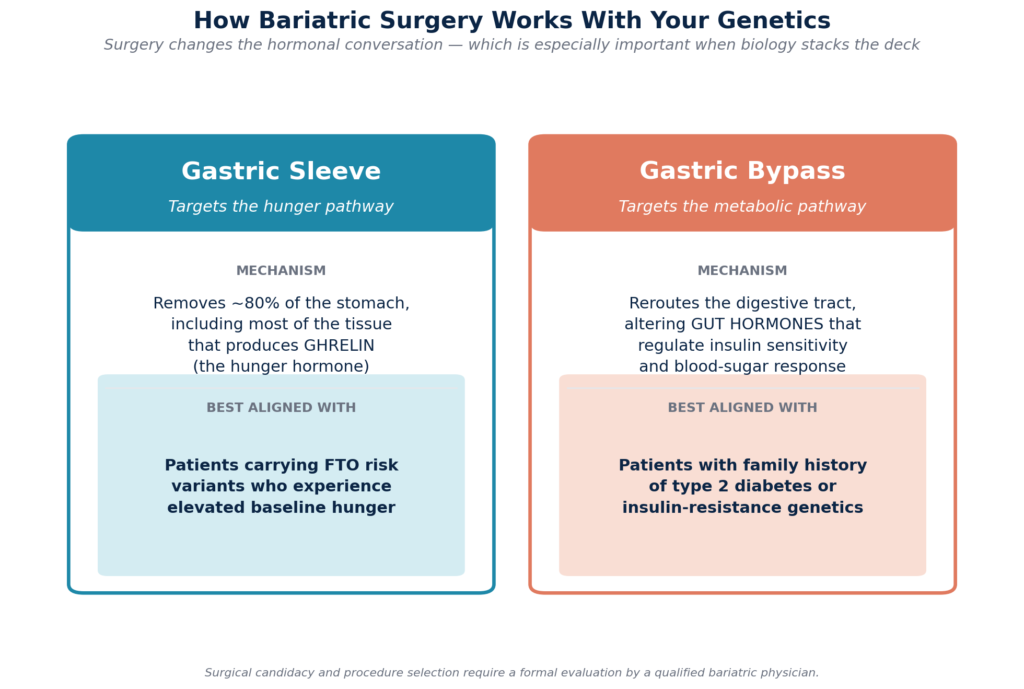
For patients whose biology makes sustained weight loss unrealistic through lifestyle alone, surgical options work with genetics rather than against them. The gastric sleeve removes the portion of the stomach that produces most of the body’s ghrelin, which can meaningfully reduce hunger in patients carrying high-risk FTO variants.
Gastric bypass goes further by altering gut hormones involved in insulin sensitivity, making it particularly useful for patients with a strong family history of type 2 diabetes. Revision procedures are available for patients whose earlier surgery has lost effectiveness.
Ethical, Practice and Professional Concerns
Genetic data should be handled responsibly. It is necessary to choose reputable testing organizations, know their privacy policies, and think critically.
A 2025 editorial in Nutrients argues that the question is no longer whether personalized nutrition is possible, but how to build the clinical-trial evidence base needed to support it at scale — while flagging meaningful ethical, economic, and implementation challenges. Genetic knowledge should be used as a complement to basic healthy habits, not a substitute for them.
Seeing a health professional is the best idea. According to the Academy of Nutrition and Dietetics, providers and registered dietitians who have been trained in nutrigenomics are able to combine the field of genetics with information regarding medical history, medications and personal health objectives. Professional nutrition associations emphasize that individualized guidance enhances safety and long-term effectiveness.
Conclusion: Redefining the Narrative of Weight Management
Sustainable weight management is no longer about coercing the body to fit into trends that aren’t meant to work for everyone. It is not just knowing how your body works, then trying to fight it. By combining DNA testing, historical family health, and evidence from scientific studies, individuals can go from confused to clear-headed.
Forget the blame game, rigid diets and endless frustration. Instead, consider research-based personalized strategies, which in some long-term trials have produced substantially greater weight loss and better cardiometabolic outcomes than one-size-fits-all diets. You are not broken. You are biologically unique. Once your strategy reflects that, it will be easier to stay consistent, predictable, and healthy. The future of weight management is no longer an issue of perfection, but rather a question of precision. And that accuracy starts with realizing your genetic blueprint.
When Genetics and Lifestyle Aren’t Enough
For some patients, genetic testing and behavioral change simply aren’t enough — the biology is too stacked, or the metabolic damage from years of struggle is too advanced. When BMI crosses clinical thresholds or obesity-related conditions have already developed, bariatric surgery in Mexico becomes a reasonable next conversation, not a last resort.
At Renew Bariatrics, we review each patient’s medical history, family history, and metabolic profile to determine whether surgery is appropriate and which procedure best fits their situation. If you’d like to see whether you qualify, our short qualification quiz is a good starting point.
Medical Disclaimer: This article is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Genetic test results, family history, and weight-management strategies should be discussed with a qualified healthcare provider. Bariatric surgery carries risks and is not appropriate for every patient; candidacy must be determined through a formal medical evaluation. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or treatment plan.
References
- Academy of Nutrition and Dietetics. (2014). Position of the Academy of Nutrition and Dietetics: Nutritional Genomics. Journal of the Academy of Nutrition and Dietetics, 114(2), 299–312. https://pubmed.ncbi.nlm.nih.gov/24439821
- AIMS Press. (2024). Unlocking weight loss potential: Investigating the impact of a nutrigenetic diet on weight loss and clinical parameters. AIMS Molecular Science, 11(1). https://www.aimspress.com/article/doi/10.3934/molsci.2024002
- Benedict, C., Axelsson, T., Söderberg, S., Larsson, A., Ingelsson, E., Lind, L., & Schiöth, H. B. (2014). Fat mass and obesity-associated gene (FTO) is linked to higher plasma levels of the hunger hormone ghrelin and lower serum levels of the satiety hormone leptin in older adults. Diabetes, 63(11), 3955–3964. https://diabetesjournals.org/diabetes/article/63/11/3955/34322/Fat-Mass-and-Obesity-Associated-Gene-FTO-Is-Linked
- Berry, S. E., Valdes, A. M., Drew, D. A., Asnicar, F., Mazidi, M., Wolf, J., et al. (2020). Human postprandial responses to food and potential for precision nutrition. Nature Medicine, 26, 964–973. https://www.nature.com/articles/s41591-020-0934-0
- Centers for Disease Control and Prevention. (2024). Risk Factors for Obesity. https://www.cdc.gov/obesity/risk-factors/risk-factors.html
- Frayling, T. M., Beaumont, R. N., Jones, S. E., Yaghootkar, H., Tuke, M. A., Ruth, K. S., et al. (2018). A common allele in FGF21 associated with sugar intake is associated with body shape, lower total body-fat percentage, and higher blood pressure. Cell Reports, 23(2), 327–336. https://pmc.ncbi.nlm.nih.gov/articles/PMC5912948
- International Sports Sciences Association. (2020). How to Develop a Workout Plan Based on Your Genetics. https://www.issaonline.com/blog/post/how-to-develop-a-workout-plan-based-on-your-genetics
- Karra, E., O’Daly, O. G., Choudhury, A. I., Yousseif, A., Millership, S., Neary, M. T., et al. (2013). A link between FTO, ghrelin, and impaired brain food-cue responsivity. Journal of Clinical Investigation, 123(8), 3539–3551. https://www.jci.org/articles/view/44403
- Lewis, C., Kelleher, I., Smyth, P. P., & Rotter, S. (2020). A comparison of a ketogenic diet with a LowGI/nutrigenetic diet over 6 months for weight loss and 18-month follow-up. BMC Nutrition, 6, 53. https://pubmed.ncbi.nlm.nih.gov/32983551
- Mansour, S., Alkhaaldi, S. M. I., Sammanasunathan, A. F., Ibrahim, S., Farhat, J., & Al-Omari, B. (2024). Precision nutrition unveiled: Gene–nutrient interactions, microbiota dynamics, and lifestyle factors in obesity management. Nutrients, 16(5), 581. https://pmc.ncbi.nlm.nih.gov/articles/PMC10935146
- MedlinePlus. (2024). What is epigenetics? U.S. National Library of Medicine. https://medlineplus.gov/genetics/understanding/howgeneswork/epigenome
- Mohammad, S., Ahmad, J., Baig, M. A., Shukla, V. K., Tanvir, I., & Zahid, T. (2020). Gene variants and pathways implicated in the development of obesity: A review. Journal of Advanced Research, 27, 95–114. https://pmc.ncbi.nlm.nih.gov/articles/PMC7296188
- National Institute of Diabetes and Digestive and Kidney Diseases. (2023). Factors affecting weight & health. https://www.niddk.nih.gov/health-information/weight-management/adult-overweight-obesity/factors-affecting-weight-health
- NIH Office of Dietary Supplements. (2024). Dietary Supplement Fact Sheets. https://ods.od.nih.gov/factsheets/list-all
- Nutrients (Editorial). (2025). Advancing personalized nutrition through genetic nutritional insights. Nutrients, 17(13), 2166. https://www.mdpi.com/2072-6643/17/13/2166
- Pickering, C., & Kiely, J. (2017). ACTN3: More than just a gene for speed. Frontiers in Physiology, 8, 1080. https://www.frontiersin.org/journals/physiology/articles/10.3389/fphys.2017.01080/full
- Tanaka, T., Ngwa, J. S., van Rooij, F. J. A., Zillikens, M. C., Wojczynski, M. K., Frazier-Wood, A. C., et al. (2013). Genome-wide meta-analysis of observational studies shows common genetic variants associated with macronutrient intake. American Journal of Clinical Nutrition, 97(6), 1395–1402. https://pmc.ncbi.nlm.nih.gov/articles/PMC3652928
- World Health Organization. (2024). Obesity and overweight. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight





